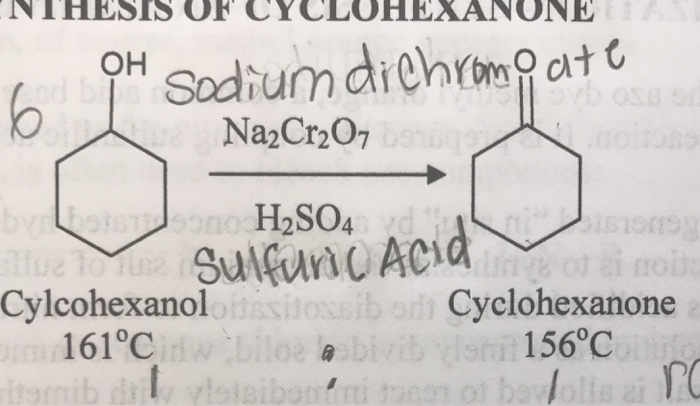
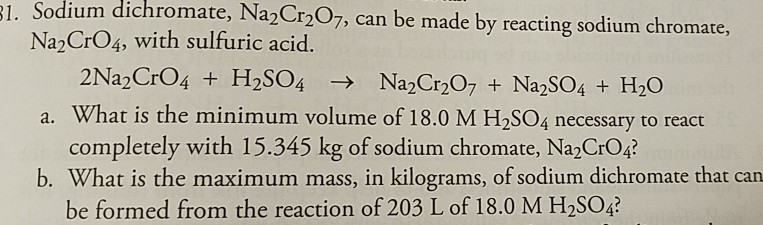SOLVED: What are the major products in each of the following oxidation with sodium dichromate in sulfuric acid? OH Leeneannenen Mett# 4 Defete diennliit OH OH OH

Wreite the balanced chemical equation of the following word equation.p Potassium dichromate + hydrochloric acid → potassium chloride + chromium chloride + water +chlorine
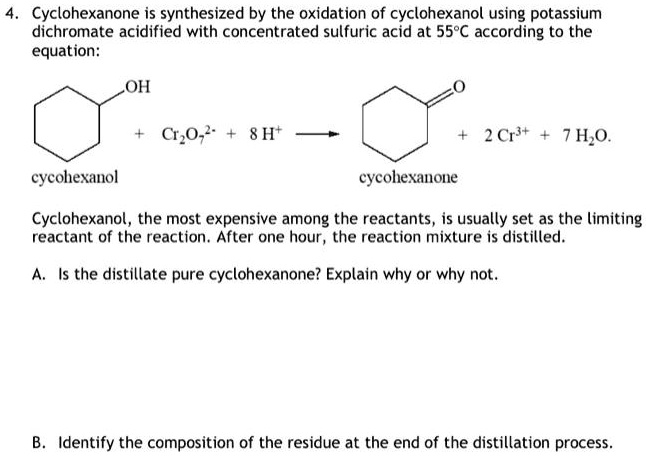
SOLVED: Cyclohexanone is synthesized by the oxidation of cyclohexanol using potassium dichromate acidified with concentrated sulfuric acid at 559C according to the equation: OH C,0z' 8 Ht 2 Crst 7 H,o. cycolexanol

When a mixture of solid sodium chloride,potassium dichromate is heated with concentrated sulphuric acid orange red vapours are formed, then compound formed is :

Mercury(II) sulfate solution for COD determination (80 g/l in potassium dichromate acidified with sulfuric acid) - Th. Geyer

The product will be an alkyl sulphonate , R-SO3H mixture of sodium dichromate and sulphuric acid will not oxidize a primary alcohol

formula of = Potassium dichromate + Sulphuric acid = Potassium sulphate + Chromium sulphate + water + - Brainly.in
IGCSE Chemistry 2017: 4.31C: Know that Ethanol can be Oxidised by: Burning in Air or Oxygen (Complete Combustion), Reaction with Oxygen in the Air to Form Ethanoic Acid (Microbial Oxidation), Heating with