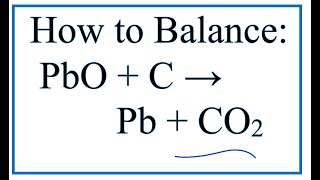
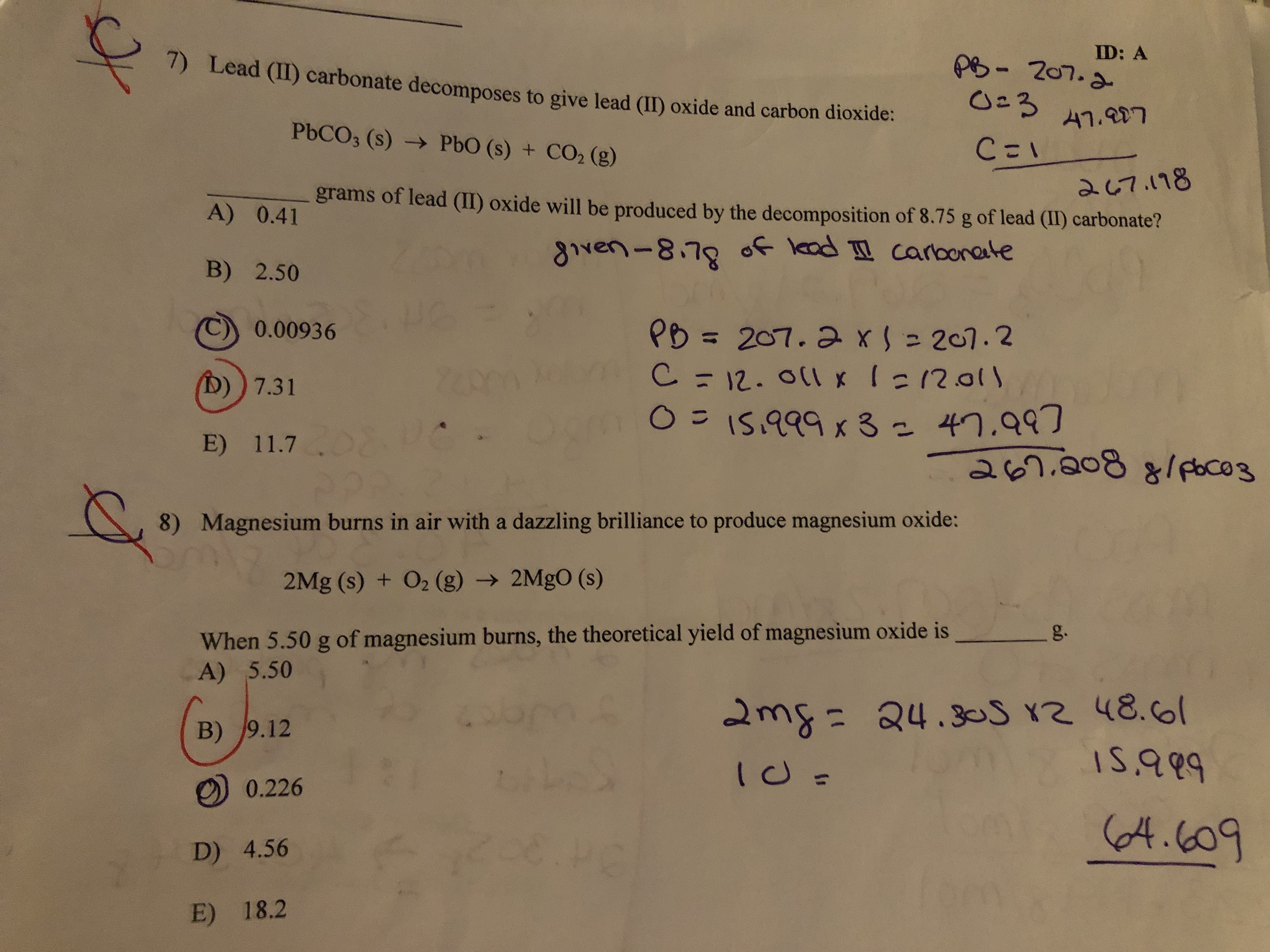Which of the statements about the reaction below are incorrect ? 2PbO(s) + C(s) rarr 2Pb(s) + CO(2)(g) (a) Lead is getting reduced. (b) Carbon dioxide is getting oxidised. (c) Carbon is
18 Which of the statements about the reaction below are incorrect? 2PbO(s)+C(s)→ 2Pb(s)+CO2(g) (a) Lead is getting reduced (b) Carbon Dioxide is getting oxidised (c) Carbon is getting oxidised (d) Lead oxide
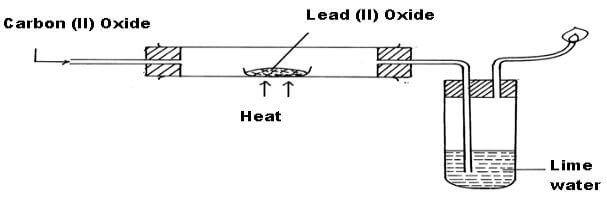
The apparatus shown below was used to investigate the effect of Carbon (II) Oxide on Lead (II) Oxide. - EasyElimu Questions and Answers
A student was trying to extract the metals from lead oxide and aluminium oxide. She heated each oxide with carbon in a fume cupb

What will be the product when lead oxide reacts with carbon i thought carbon - Science - Acids Bases and Salts - 12499069 | Meritnation.com
![For the given reaction,\\[2PbO + C \\to Pb + C{O_2}\\] name the oxidized substance, reduced substance, reducing agent, and oxidizing agent in this reaction. For the given reaction,\\[2PbO + C \\to Pb + C{O_2}\\] name the oxidized substance, reduced substance, reducing agent, and oxidizing agent in this reaction.](https://www.vedantu.com/question-sets/407a8186-d96f-4ae5-9b1b-a35a89f410b32918307568284777948.png)
For the given reaction,\\[2PbO + C \\to Pb + C{O_2}\\] name the oxidized substance, reduced substance, reducing agent, and oxidizing agent in this reaction.
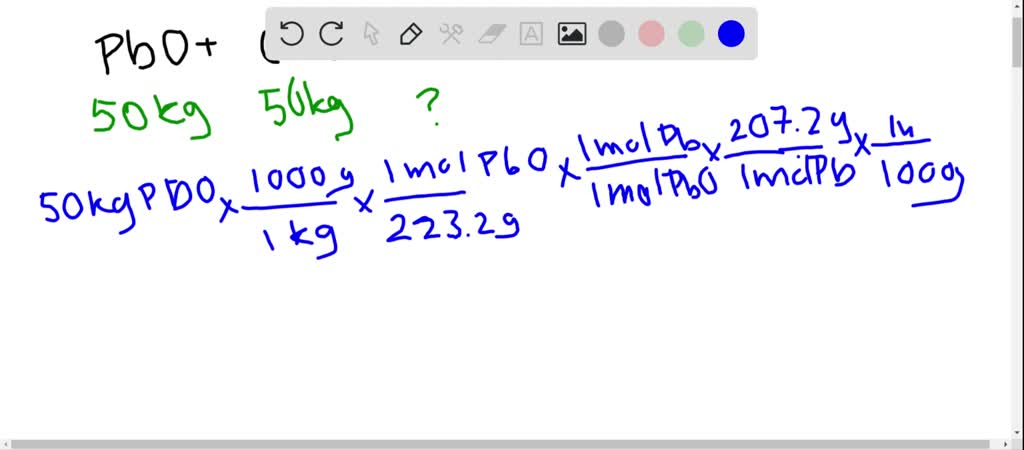
SOLVED:Lead(II) oxide from an ore can be reduced to elemental lead by heating in a furnace with carbon. PbO(s)+C(s) →Pb(l)+CO(g) Calculate the expected yield of lead if 50.0 kg of lead oxide

Preparation of High Purity Lead Oxide from Spent Lead Acid Batteries via Desulfurization and Recrystallization in Sodium Hydroxide | Industrial & Engineering Chemistry Research

Which of the statements about the reaction below are incorrect ? 2PbO(s) + C(s) → 2Pb(s) + CO2(g) (a) Lead is getting reduced.(b) Carbon dioxide is getting oxidised.(c) Carbon is getting oxidised.(d)

Marie mixed 5g of carbon with 5g of lead oxide. she heated the mixture strongly for 15 minutes in a - Brainly.com

SOLVED: Lead (II) carbonale decomposes t0 give lead (IIl) oxide and carbon dioxide: PbCO3 (s) + PbO (s) COz (g) grams of lead (II) oxide will be produced by the decomposition of